- The Zeroth Law: The Thermometer’s Logic
- The First Law: The Cosmic Ledger
- The Second Law: The Arrow of Time
- The Third Law: The Absolute Stop
- The Heat Death: The Ultimate Buzzkill
- Focus on Language
- Critical Analysis
- Let’s Discuss
- Fantastic Guest: An Interview with Ludwig Boltzmann
- Let’s Play & Learn
- Check Your Understanding (Quiz)
If you have ever stared helplessly at a cup of coffee that went cold while you were distracted by an email, or if you have ever cleaned your apartment on a Saturday morning only to find it looking like a bomb site by Tuesday evening, you have engaged in a personal battle with Thermodynamics. And let me tell you right now: you lost. You will always lose.
Thermodynamics is not just a dusty chapter in a physics textbook involving steam engines and pistons. It is the operating system of reality. It explains why time moves forward, why engines get hot, why we eat food, and ultimately, how the universe will end. It is the study of energy and how it moves. And unlike the laws of traffic or the laws of etiquette, the laws of thermodynamics are strictly enforced by the cosmos itself. There are no loopholes. There is no appeal court.
We tend to think of energy as this mystical “vibe” or the thing we lack on Monday mornings, but in physics, energy is the currency of existence. Thermodynamics is the accounting system that tracks every single penny of that currency. And the audit is terrifying.
The Zeroth Law: The Thermometer’s Logic
Defining the Rules of the Game
Before we get to the heavy hitters, we have to start with the Zeroth Law. Yes, it is called the Zeroth Law because scientists figured out the First and Second laws before realizing they missed a prerequisite. It’s like realizing you forgot to plug in the console after you’ve already bought the game.
The Zeroth Law states: “If two systems are each in thermal equilibrium with a third system, they are in thermal equilibrium with each other.”
Sounds dry, right? But let’s translate that. This is the law that allows thermometers to exist. If your thermometer (System A) is the same temperature as a glass of water (System B), and that glass of water is the same temperature as the air in the room (System C), then the thermometer is also the same temperature as the room. It defines temperature not as a subjective feeling—”ooh, it’s chilly”—but as a measurable, transitive property of matter. It establishes the baseline: energy wants to flow until everything is equal. It wants balance. And as we will see, the universe’s obsession with balance is what eventually kills everything.
The First Law: The Cosmic Ledger
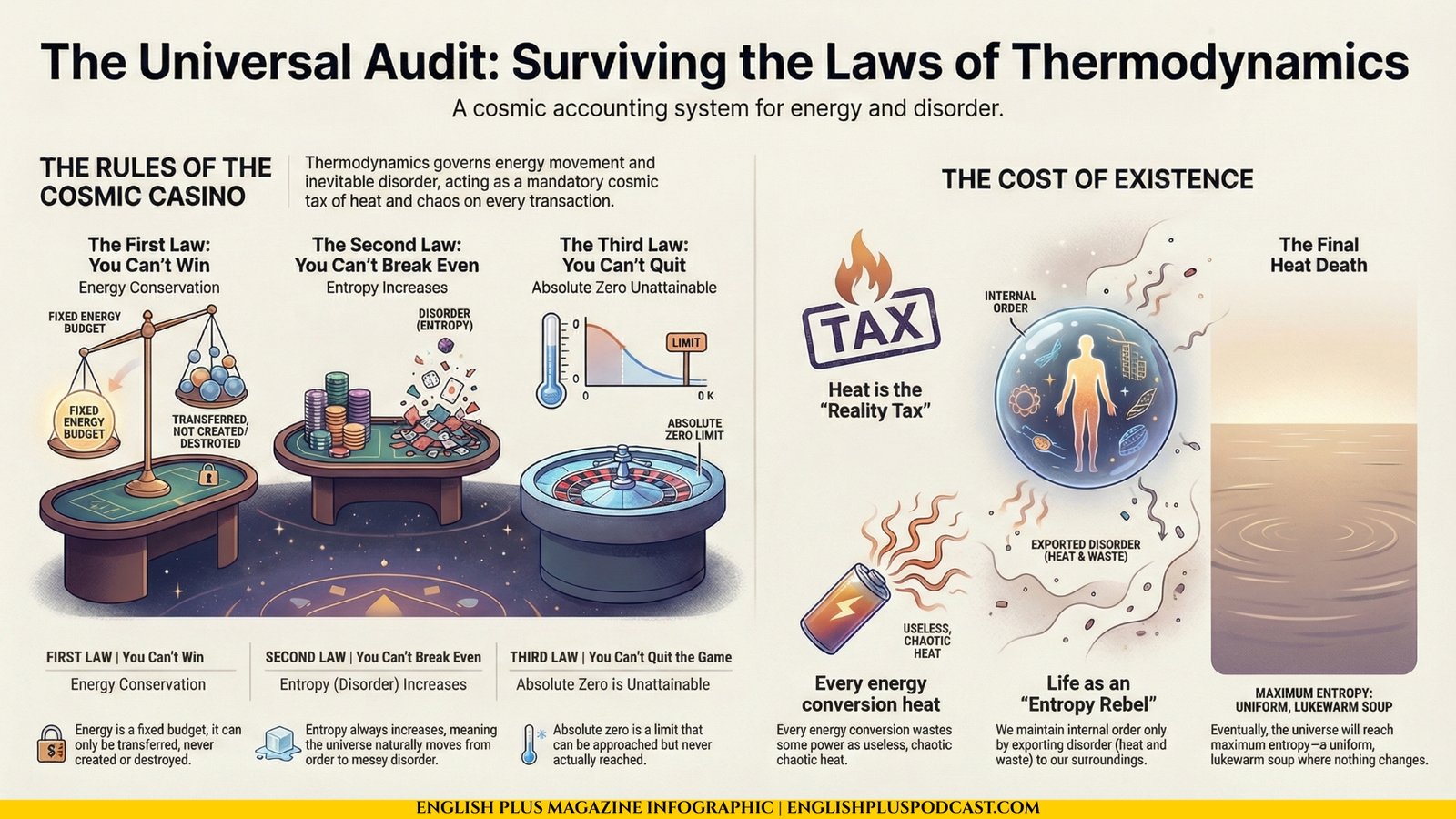
You Can’t Win
The First Law of Thermodynamics is the Law of Conservation of Energy. It is the bedrock of physics. It states simply: Energy cannot be created or destroyed, only transferred or changed from one form to another.
In casino terms: You can’t win.
You cannot get something for nothing. There is no such thing as a free lunch, and there is certainly no such thing as a perpetual motion machine. Every machine, every organism, every star is just a transformer. You eat a burger (chemical energy), and your body turns it into movement (kinetic energy) and body heat (thermal energy). You put gas in your car, and the engine turns it into motion and a whole lot of wasted heat.
This law is strangely comforting and deeply restrictive. It means the universe has a fixed budget. The total amount of energy in the universe today is exactly the same as it was at the Big Bang. It just looks different. It has spread out. We aren’t creating new light; we are just recycling old energy.
This is why those YouTube videos of “free energy” magnets spinning forever are nonsense. If a wheel is spinning, it is fighting friction. Friction generates heat. That energy has to come from somewhere. If the machine isn’t plugged in, the energy is coming from the spin itself, meaning the wheel must slow down. The First Law is the bouncer at the club of reality, checking your pockets to make sure you didn’t sneak in any extra joules.
The Second Law: The Arrow of Time
You Can’t Break Even
If the First Law says you can’t win, the Second Law delivers the knockout punch: You can’t even break even.
The Second Law of Thermodynamics is arguably the most profound and depressing law in all of science. It introduces a concept called Entropy. In simple terms, entropy is a measure of disorder, or randomness. The law states: “In an isolated system, the total entropy can never decrease over time.”
Basically, the universe has a natural tendency to move from order to disorder. Heat flows from hot to cold, never the reverse. If you put an ice cube in hot coffee, the heat flows into the ice, melting it. You never see a lukewarm cup of coffee suddenly decide to separate into boiling liquid and a frozen cube. That would not violate the First Law (energy would still be conserved), but it violates the Second Law.
This is why your room gets messy. There are a million ways for your socks to be scattered on the floor (high entropy/disorder), but only one way for them to be folded neatly in the drawer (low entropy/order). Random chance favors the mess. To create order, you have to input energy. You have to expend effort to fold the socks. But—and here is the kicker—by expending that effort, you are burning calories, generating body heat, and exhaling carbon dioxide. You are creating more disorder in the universe (via heat and waste) than the order you created in the drawer.
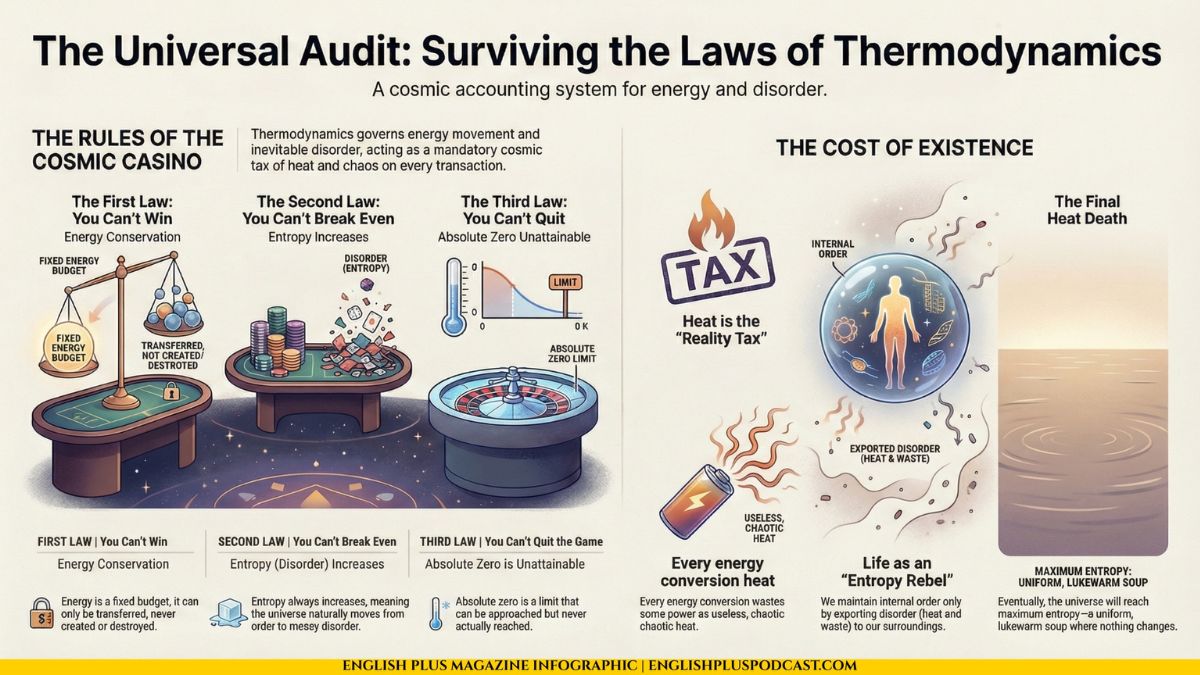
The Tax of Reality
Every time energy is converted—from coal to electricity, from electricity to light, from a burger to a heartbeat—some of that energy is wasted as heat. Heat is the graveyard of energy. It is disordered, chaotic vibration of molecules. Once energy turns into low-grade heat, it is very hard to do anything useful with it.
Think of it like currency exchange. Every time you change dollars to euros, the bank takes a fee. If you keep switching back and forth, eventually you have zero money left, and the bank has it all. In the universe, “heat” is the bank fee. We are slowly converting all the useful, concentrated energy (stars, batteries, fuel) into useless, spread-out heat.
This leads to the concept of the “Arrow of Time.” Newton’s laws of motion work backward and forward. If you play a video of billiard balls colliding in reverse, it still looks like physics. But if you play a video of an egg smashing on the floor in reverse, it looks wrong. You know it’s fake. Why? Because of Entropy. We can tell the past from the future because the future is the direction where things are messier.
The Third Law: The Absolute Stop
You Can’t Quit the Game
The Third Law of Thermodynamics deals with the absolute limit of cold: Absolute Zero (-273.15°C or 0 Kelvin). It states that the entropy of a perfect crystal at absolute zero is exactly zero.
Basically, if you could stop all atomic motion, you would have perfect order. No disorder. No entropy. But here is the catch: You can’t get there.
You can get incredibly close. We have cooled atoms to fractions of a degree above absolute zero. But you can never reach the true zero. To cool something, you have to remove heat. But the process of removing heat requires work, and doing work generates heat nearby. It’s a thermodynamic Zeno’s Paradox. You can get halfway there, and halfway again, but you can never step off the playing field. The atoms will always shimmy, just a little bit.
The Heat Death: The Ultimate Buzzkill
A Universe of Lukewarm Soup
If we follow these laws to their logical conclusion, we arrive at a rather somber destination. Since entropy is always increasing, and useful energy is always dissipating into heat, eventually, there will be no useful energy left.
Stars will burn out. Black holes will evaporate. The universe will continue to expand and cool. Eventually, the entire cosmos will reach a uniform temperature just above absolute zero. No energy will flow because everything will be the same. No life can exist. No machines can run. Time itself effectively stops because nothing changes.
This is called the “Heat Death of the Universe.” It is the ultimate messy room. Maximum entropy. Total equilibrium.
Life as a Rebel
But wait. If the universe loves disorder, why are you here? You are a highly ordered, complex structure. A tree is ordered. A city is ordered. Doesn’t life violate the Second Law?
No. Life is a loophole exploiter. We are open systems. We are not isolated. We survive by feeding on low-entropy energy (sunlight, food) and excreting high-entropy waste (heat, poop). We maintain our internal order by exporting disorder to our surroundings. We are locally reversing entropy, but globally increasing it.
Think of a refrigerator. It makes the inside cold (ordered) by pumping heat out the back (disorder). If you feel the back of your fridge, it’s hot. Life is just a very complicated refrigerator that thinks. We are surfing a wave of chaos, building sandcastles while the tide comes in. And while the tide will eventually win, the sandcastles are pretty magnificent in the meantime.
Focus on Language
Let’s shift gears and look at the language we used to describe these heavy concepts. Science writing can be dry, so using rich, metaphorical language is key to making it stick. I want to look at the word Dissipate. I used this when talking about heat. To dissipate means to scatter or disappear, but usually in a way that involves spreading out until something vanishes. Smoke dissipates. Your savings might dissipate if you aren’t careful. In thermodynamics, energy dissipates into heat—it’s not gone (First Law), but it’s spread out so thin it’s useless. You can use this in real life: “Her anger began to dissipate after she ate lunch.”
Then we have Equilibrium. This is the state of balance. In physics, it’s when temperature equalizes. But socially, it means a state of calm or stability. If your life feels chaotic, you might say, “I’m just trying to find some equilibrium between work and sleep.” It’s a sophisticated way of saying “balance.”
I mentioned Isolated System. In physics, this is a system where nothing comes in or out. A thermos is a decent attempt at an isolated system. The universe is the ultimate isolated system. In conversation, you might say, “You can’t treat this problem as an isolated system; everything is connected.” It implies that outside factors matter.
We talked about Chaos. We usually think of chaos as people running around screaming, but in science, chaos refers to randomness and disorder—high entropy. You can use this to describe your desk: “My filing system is pure chaos.” It implies a lack of structure or predictability.
Let’s look at Irreversible. The breaking of an egg is irreversible. You can’t un-break it. The Second Law defines the irreversible flow of time. In life, we make irreversible decisions. “Signing that contract was an irreversible step.” It adds a weight of permanence that words like “permanent” sometimes lack.
I used the phrase Perpetual Motion. This refers to a machine that moves forever without an energy source. It’s impossible. But we use it metaphorically to describe people with high energy. “My toddler is a perpetual motion machine.” It’s a hyperbole, obviously, but it paints a vivid picture of non-stop action.
We discussed the Macrostate and Microstate implicitly with the socks analogy. A “macrostate” is the big picture (Messy Room). The “microstate” is the specific arrangement of every single sock. There are many microstates that look like “messy,” but few that look like “clean.” You can use “macro” and “micro” to talk about economics or planning. “Let’s look at the macro view before we get bogged down in the micro details.”
Then there is Finite. The energy in the universe is finite. It has a limit. Our time is finite. This is a great word to remind people of scarcity. “I have a finite amount of patience for this nonsense.” It sounds much more authoritative than “I don’t have much patience.”
I used the word Spontaneous. In chemistry and physics, a spontaneous reaction is one that happens on its own without needing a push (like ice melting in warm air). In life, spontaneous means impulsive or unplanned. “We took a spontaneous trip to the beach.” It’s the same root concept—happening naturally without external force.
Finally, Conservation. We talked about the Conservation of Energy. To conserve is to protect from loss. We talk about nature conservation. But you can also conserve your strength. “I’m staying home tonight to conserve my energy for the big presentation.” It frames your laziness as a strategic resource management decision.
Now, for the speaking section. I want to challenge you on the concept of Analogy. Thermodynamics is abstract, so we understand it through comparison (the bank, the messy room, the casino).
Your assignment: I want you to look around the room you are in right now. Find something that is “broken” or “messy” or “decaying.”
I want you to describe it using the word Entropy.
Instead of saying “My kitchen is dirty,” say: “The entropy in my kitchen has reached a critical level.”
Instead of saying “My phone battery is dead,” say: “My phone has reached thermodynamic equilibrium with the charger.”
Record yourself giving a 30-second “Scientific Status Report” of your life using these physics terms. It makes the chaos of life sound intentional and smart. “I am not lazy; I am simply obeying the Second Law of Thermodynamics.”
Critical Analysis
We have presented Thermodynamics as the ultimate, unbreakable law of the universe. The “Heat Death” narrative is the standard scientific dogma. But let’s play the devil’s advocate. Is the Second Law really as absolute as we say? Or are we missing something in the fine print?
First, let’s look at the Statistical Nature of the Law. The Second Law is not a “law” in the same way gravity is. It is a statistical probability. It is overwhelmingly likely that entropy will increase, but it is not theoretically impossible for it to decrease by pure chance.
There is a concept called the Poincaré Recurrence Theorem. It suggests that if you have a finite system with finite energy (like a box of gas… or the universe), given an infinite amount of time, the particles will eventually, randomly, return to their original state. That means, if you wait long enough (trillions upon trillions of years), your shattered egg could spontaneously reassemble itself just by the random motion of atoms. It’s incredibly unlikely, but not impossible. By presenting the Second Law as absolute, we might be ignoring the bizarre possibilities of infinity.
Secondly, let’s challenge the definition of “Disorder.” We equate entropy with “messiness.” But this is a human-centric simplification. In Information Theory (the math behind computers), entropy is actually a measure of information. A perfectly ordered string of letters (AAAAAAA) has low entropy and contains very little information. A random string (XJ9#mK2) has high entropy and high information density.
So, is the universe getting “messier,” or is it getting more complex? As stars die and spread their dust, that dust forms planets, which form life, which forms brains. The local complexity is rising. By focusing only on the “Heat Death,” we might be engaging in a form of nihilism that ignores the incredible creativity of the dissipative process. The universe dies, yes, but look at what it builds on the way down.
Third, we must consider Maxwell’s Demon. This was a thought experiment by physicist James Clerk Maxwell. He imagined a tiny demon controlling a door between two gas chambers. The demon lets fast molecules go one way and slow molecules go the other, creating a temperature difference without expending energy. This would violate the Second Law.
For a century, this troubled physicists. Eventually, they realized the demon would need to measure the speed of the molecules, and the act of storing that information increases entropy. But this highlights a deep connection between Physics and Information. Are the laws of thermodynamics actually laws of data processing? Some modern physicists argue that the universe is essentially a computer, and entropy is the cost of erasing memory. If we view reality as computation, the “Heat Death” isn’t a death; it’s just the program finishing its run.
Finally, let’s talk about Open vs. Closed Systems. We apply the Second Law to the universe because we assume the universe is an isolated system. But what if it isn’t? What if our universe is part of a multiverse, leaking energy in or out? If the system isn’t closed, the Second Law doesn’t apply globally. We are making a massive assumption about the container of reality based on what we can see from our tiny corner.
So, while you definitely can’t build a perpetual motion machine in your garage, the ultimate fate of the cosmos might not be as settled as the “Heat Death” theory suggests.
Let’s Discuss
Here are five questions to get you thinking about the philosophical implications of heat and chaos.
1. If the universe is destined for Heat Death, does anything we do matter?
This is the ultimate existential question. If all monuments crumble and all stars fade, is legacy a lie? Or does the temporary nature of beauty make it more valuable, like a flower that blooms for only one day?
2. Can we ever truly “waste” energy?
The First Law says energy is never destroyed. So when we leave the lights on, we aren’t destroying energy, we are just converting it to heat. Is “waste” a physical concept, or just a human economic concept?
3. Is life a struggle against physics?
We talked about life being a “rebel.” Do you view your own life as a constant effort to create order out of chaos? Is cleaning your room a spiritual act of defying the Second Law?
4. If time is just the increase of entropy, would time run backward in a shrinking universe?
Stephen Hawking once entertained this idea. If the Big Bang reversed into a Big Crunch, and the universe got more ordered, would we remember the future and forget the past? What is the connection between “memory” and “entropy”?
5. Would you want to live in a world without entropy?
A world without entropy is a world without change. Nothing breaks, but nothing grows. Time stands still. Is the pain of decay a fair price to pay for the joy of progress?
Fantastic Guest: An Interview with Ludwig Boltzmann
Danny: Welcome back to the Fantastic Guest segment. We’ve spent some time talking about the laws of the universe—specifically, the laws that say we can’t win, can’t break even, and definitely can’t quit. We’ve talked about entropy, the relentless march of disorder, and the eventual Heat Death of everything we love. Cheerful stuff.
To discuss this, I didn’t want a steam engine mechanic. I wanted the man who looked into the abyss of probability and saw the atoms dancing. He is the father of Statistical Mechanics, the man who proved that heat is just motion, and the genius who gave us the formula for entropy—which, by the way, is carved on his tombstone. He spent his life fighting the scientific establishment to prove that atoms exist, and tragically took his own life just before the world admitted he was right. Please welcome the Austrian visionary, Ludwig Boltzmann. Herr Boltzmann, welcome.
Boltzmann: You are very loud. And your studio… it is very ordered. Low entropy. It makes me suspicious.
Danny: I cleaned up for you. I didn’t want to offend the master of disorder.
Boltzmann: You misunderstand. I do not love disorder. I simply recognize its inevitability. A clean room is a statistical improbability. You are fighting the universe just by organizing those papers, Danny. It is a noble, futile war.
Danny: That’s the theme of our issue today: “The Futile War.” Ludwig—can I call you Ludwig?
Boltzmann: You may. Though in Vienna, we were more formal. But I am a ghost, so titles matter little.
Danny: Ludwig, you are famous for linking the behavior of atoms to the concept of entropy. But back in the late 19th century, people didn’t even believe atoms were real. Ernst Mach famously asked you, “Have you ever seen one?” It must have been maddening.
Boltzmann: “Maddening” is a mild word. It was psychological torture. Imagine knowing how a clock works—seeing the gears in your mind, calculating their movements perfectly—and having everyone around you scream, “There are no gears! Time is magic!”
Mach… Ostwald… they were brilliant men, but they were stubborn. They thought energy was a continuous fluid. I told them, “No! It is packets! It is particles bouncing like billiard balls!” They laughed. They called me a materialist. They said I was inventing fairy tales.
And now? I look at your world. You split the atom. You build bombs with it. You build power plants. I was right. But being right is a lonely comfort when you are dead.
Danny: That’s the tragedy of your life. You died in 1906. Einstein proved the existence of atoms explicitly in 1905 with Brownian motion. You missed the victory parade by that much.
Boltzmann: Fate has a cruel sense of humor. It is high entropy. A chaotic twist. If I had waited… perhaps I would have seen the dawn. But the darkness was very heavy then. I felt that physics was moving backward, away from truth. I felt I was the last guardian of the atom. It is a heavy burden to carry a microscopic universe on your back.
Danny: Let’s talk about that burden. You formulated the equation S = k log W. It connects the macro world (temperature, heat) with the micro world (atoms, probability). For the non-physicists listening, how would you explain “W”?
Boltzmann: Ah, the W. Wahrscheinlichkeit. Probability. The number of ways.
Think of it this way, Danny. You have a deck of cards. If you throw them in the air, what is the likelihood they land in a perfect stack, in numerical order?
Danny: Basically zero.
Boltzmann: Exactly. There is only one way for them to be perfectly ordered. That is a low W.
But how many ways are there for them to be scattered all over the floor? Billions. Trillions. That is a high W.
Entropy (S) is just a measure of that probability. Nature prefers the high W simply because there are more of them. The cards scatter not because a demon pushes them, but because there are more ways to be scattered than to be stacked.
It is not a force. It is just… statistics. The universe is a gambler, and the house always wins because the house has more winning tickets.
Danny: That’s a terrifyingly simple explanation. It means disorder isn’t a mistake; it’s just the default setting because it’s statistically more likely.
Boltzmann: Precisely. You exist in a highly unlikely state, Danny. Your body is a complex arrangement of atoms that requires constant energy to maintain. You are a statistical anomaly. You should be dust. The fact that you are not dust is a temporary victory against the odds.
Danny: Thanks, I feel very lucky to be non-dust today. But this leads to the scary part: The Arrow of Time. We talked about how you can’t un-break an egg. Your theory says it’s not impossible, just unlikely.
Boltzmann: Yes. This is where I upset people.
According to Newton, time can go backward. Atoms can bounce left or right. If you reverse the film, the physics still works. So why do we never see the egg un-break?
My answer was the H-theorem. I showed that if you start with an ordered state, the system will almost always evolve toward a disordered state, simply because there are more disordered states.
But… “almost always” is not “always.”
If you wait long enough—an eternity of eternities—the atoms of the broken egg will randomly bump into each other in just the right way to reform the egg. It will happen. The math demands it. But the time required is so vast that the universe will likely end before it happens.
So, for all practical purposes, time flows one way. Toward the mess.
Danny: That connects to the Poincaré Recurrence Theorem we discussed in the “Critical Analysis.” But Ludwig, this idea—that the universe is just shuffling from order to disorder—leads to the Heat Death. A universe of lukewarm soup. Did that thought haunt you?
Boltzmann: It haunted me deeply. If the Second Law is true everywhere, then the universe must be winding down. Like a clock that was wound up once at the beginning and is slowly ticking toward a stop.
This implies a beginning—a low-entropy state—and a tragic end. A cold, dead silence.
For a man who loved beauty, who loved music—I played the piano, you know—the idea that the final note of the symphony is just… static? It is depressing. It makes one question the point of the struggle. Why build the house if the wind will inevitably blow it down?
Danny: And what is the answer? Why build the house?
Boltzmann: Because we are here now. The low entropy exists now.
I loved the opera. I loved a good dinner. The fact that the dinner will eventually be… well, processed… does not make it less delicious in the moment.
We are the beneficiaries of a cosmic accident. The universe started in a state of high order (the Big Bang). We are riding the slide down. The slide is fun, Danny. Even if it ends in the mud.
Danny: “The slide is fun.” I like that.
I want to ask you about your rival, James Clerk Maxwell. He came up with “Maxwell’s Demon”—a little creature that could sort atoms and violate the Second Law. Did you think his demon was a threat to your theory?
Boltzmann: Maxwell was a genius. His demon was a clever puzzle.
He imagined a being that could open a door to let fast atoms through and close it for slow ones, creating heat without work.
But Maxwell forgot the cost of knowing. To sort the atoms, the demon must observe them. He must measure their speed. He must store that information in his brain.
Information is physical, Danny. A memory is a rearrangement of atoms in the brain. The act of measuring increases the entropy of the demon. So, the total entropy of the system (box + demon) still goes up.
The demon sweats. He burns calories to do the sorting. The Second Law is safe. You cannot cheat the house by being smart. Being smart costs energy.
Danny: That’s a brilliant connection to the modern idea that “Information is Physics.”
Let’s get a bit weird. Have you heard of the “Boltzmann Brain”?
Boltzmann: I have heard what the philosophers did to my name.
Danny: For the audience: The Boltzmann Brain theory suggests that since low-entropy states (like a human brain) can randomly fluctuate out of chaos given enough time, it is statistically more likely for a single brain to pop into existence in the void, fully formed with false memories, than for an entire universe to evolve to create us.
So, Ludwig… am I real? Or am I just a brain floating in space hallucinating this interview?
Boltzmann: You ask me this? I am a ghost talking to a man here. The layers of reality are already very thin, Danny.
But to answer you: The Boltzmann Brain paradox is a reductio ad absurdum. It shows that if the universe is infinite and eternal, probability gets very strange.
If you rely only on random fluctuation, yes, a single brain is more likely than a whole galaxy. But we observe a whole galaxy. We observe a consistent past. We observe dinosaur bones.
This implies that we are not the result of a random fluctuation in a dead universe. We are the result of a specific process—evolution—that started from a low-entropy Big Bang.
So, you are likely real. Or at least, you are as real as I was. Which, given my mental state at the end, is perhaps not the best reassurance.
Danny: I’ll take “likely real.”
I want to talk about “Life as a Rebel.” We described life as a “loophole exploiter” in the article. We decrease our own entropy by increasing the entropy of our surroundings. We eat the burger, we create heat.
Do you view life as a defiance of your law, or the ultimate expression of it?
Boltzmann: Life is the most efficient way to increase entropy.
Think about it. A rock sits in the sun. It gets warm. It radiates heat slowly.
A plant sits in the sun. It captures the light, grows, dies, rots. The rotting releases heat.
An animal eats the plant, runs around, breathes, burns energy rapidly.
We are engines of dissipation. Nature built us because we are very, very good at turning useful energy into useless heat. We are not rebels, Danny. We are the Second Law’s best employees. We accelerate the destruction of the universe, and we call it “civilization.”
Danny: Wow. That is a dark spin. We aren’t creating order; we are just really efficient trash compactors for the universe.
Boltzmann: Do not be offended. To facilitate the flow of energy is a noble purpose. We are the dancers in the ballroom of thermodynamics. We keep the music playing a little faster.
Danny: Ludwig, you were known for being bipolar, or at least struggling with severe mood swings. You called them your “periods of misery.” Do you think your mental state allowed you to see the world differently? Did the chaos in your mind help you understand the chaos of matter?
Boltzmann: It is possible. When one’s mind is a storm, one learns to look for patterns in the rain.
My colleagues saw the world as a solid, static painting. I saw it as a swirling cloud. I felt the vibration of the atoms because I felt the vibration of my own nerves.
To propose that all solidity is an illusion—that the table you sit at is mostly empty space and jittery particles—requires a mind that is willing to let go of “common sense.” My mind was very loose with common sense. It allowed me to see the unseen. But it also stripped away the protective illusions that keep men sane.
Danny: “Protective illusions.” Like the illusion that things will last?
Boltzmann: Yes. The illusion of permanence. We build statues of bronze and think, “This is forever.” I look at the statue and I see the oxidation. I see the metal fatigue. I see the atoms migrating. I see the dust it will become.
It makes it hard to be proud of anything. But it also makes you cherish the moment. If the statue is dust tomorrow, then I must admire the light on it today.
Danny: That brings us back to the crossword puzzle definition of “Finite.” We talked about how scarcity gives value.
You mentioned you played piano. Who was your favorite composer?
Boltzmann: Beethoven. Of course. He understood the struggle. He understood the violence of emotion. Mozart is too polite. The universe is not polite. The atoms collide with violence. Beethoven sounds like atoms colliding.
Danny: We actually interviewed Beethoven in the last issue. He said he liked heavy metal.
Boltzmann: This does not surprise me. I would have liked to hear this “heavy metal.” Is it high entropy music?
Danny: Extremely high entropy. Lots of distortion.
Ludwig, I have to ask about the “Zeno’s Paradox” of the Third Law. Absolute Zero. We can’t reach it. Why does the universe forbid us from stopping?
Boltzmann: Because to stop is to know everything.
If an atom stops, its position is fixed. Its momentum is zero. You know exactly where it is and how it moves (or doesn’t).
But nature hates certainty. Even before Heisenberg found his Uncertainty Principle, I suspected this. Nature requires a wiggle. The wiggle is the life. If you reach absolute zero, you have removed the probability. You have collapsed the deck of cards into a single block. The game ends. The universe wants the game to continue.
Danny: I love that. “The wiggle is the life.”
So, if you could go back to 1890, to the halls of the University of Vienna, with what you know now… what would you say to Ernst Mach?
Boltzmann: I would not say anything. I would hand him a Geiger counter and a piece of Uranium.
I would let him hear the click… click… click… of the atoms decaying.
And I would say, “Ernst, that is the sound of your philosophy crumbling.”
But I would be kind. He was a great thinker, even if he was wrong. Being wrong is part of the scientific process. It increases the entropy of knowledge, which eventually leads to the order of truth.
Danny: You are surprisingly forgiving for a man whose life was made miserable by these arguments.
Boltzmann: Death gives you perspective, Danny. The arguments of academia seem very small when you are floating in the eternal void.
We were all just trying to describe the elephant in the dark. I touched the trunk. He touched the tail. We argued. But it was the same elephant.
Danny: Before we wrap up, I want to give you a chance to redeem the “Messy Room” analogy. We use it all the time to explain entropy to kids. Is it a good analogy, or does it make you cringe?
Boltzmann: It is… adequate. But it is lazy.
The messy room implies that human “tidiness” is the preferred state of the universe. The universe does not care if your socks are folded.
A better analogy is perfume. Open a bottle of perfume in a corner. The molecules spread out to fill the room. They never rush back into the bottle.
Why? Not because they like the room better. But because there is more “room” in the room than in the bottle.
It is about freedom. Entropy is the measure of freedom. The atoms want to explore every possible location. They want to be free.
So, tell your readers: Your messy room is just your socks exercising their freedom.
Danny: “Entropy is the measure of freedom.” That is a beautiful reframe. It makes the heat death sound like a liberation.
Boltzmann: Perhaps it is. When the energy is spread perfectly evenly, every part of the universe is equal. No hot, no cold. No rich, no poor. Just… light. A democracy of photons.
Danny: Ludwig, this has been deep, dark, and oddly uplifting.
One final question. You famously said, “Bring forth the numbers.” You trusted math above all else. In a world of “fake news” and “alternative facts,” do you still believe math is the ultimate truth?
Boltzmann: Math is the only language that does not lie. Men lie. Words lie. Even senses lie. But an equation? S = k log W. It is true in Vienna, it is true on Mars, it is true at the end of time.
Trust the numbers, Danny. But remember to check your assumptions.
Danny: I will. And I’ll try to embrace the chaos of my desk.
Ludwig Boltzmann, thank you for joining us from the other side of the probability curve.
Boltzmann: It was a high probability event that I would enjoy this. Auf Wiedersehen.








0 Comments